Working principle & Instrumentation
 |
| XPS instrumentation |
XPS (photoelectron spectroscopy) is a surface-sensitive technique used to analyze the elemental composition, chemical state, and electronic state of elements on the surface of a material.
In Xps photoelectrons emitted from material as function of their binding energy in the sample, allowing for the identification of elements and their chemical states. It is highly sensitive to the top few nanometers of a material, making it valuable for surface analysis. XPS provide more athentic data than EDX.
XPS instrument typically consists of an X-ray source,usually Mg kα hv=1253.6ev line width = 0.7ev Al kα hv=1486.6ev line width=0.85ev, an hemisphere electronenergy analyzer outer negative and inner positve lining to intact electron in center, and a detector. X-rays are used to excite electrons from the sample, and the emitted photoelectrons are analyzed based on their kinetic energy (KE). XPS involves irradiating a material's surface with X-rays, which causes the emission of photoelectrons from the inner electron shells of atoms. The kinetic energy and number of emitted electrons are measured through detecter or analyser to determine the binding energy (BE) and abundance of each element providing information about the material's elemental composition electronic structure density and chemical states. Xps measures mostly top 10nm and provide surface information. Total energy of the system is hv=KV+BE+Φ si is work function, Xps can be used in line profiling of the elemental composition across the surface or
in depth profiling when pair with ion beam itching.
Sample Preparation Samples should be clean free of contamination. Conductive samples are preferred to avoid charging effects. Additionally, samples may need to be rotated during analysis to ensure a representative surface is probed.
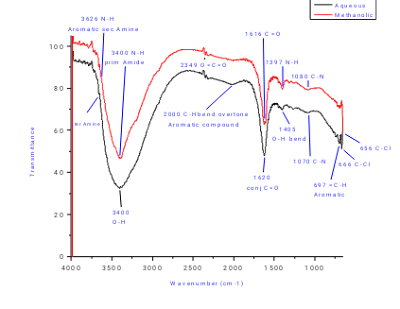
Results Interpretation XPS spectra show peaks corresponding to different elements, the position of these peaks provides information about the chemical state of the elements. The intensity of the peaks is proportional to the elemental concentration. Different peaks are making in single graph shows oxidation state known as deconvolution, when one peak get difused new one is start making this
type of peaks called satellite peaks indicate the change in the oxidation state.
 |
| XPS spectrum |






.jpeg)





.jpeg)
